Question:
Will the glue, water and liquid starch form a solution, suspension or colloid?
Hypothesis:
We think that the glue, water and liquid starch will form a solution because the mixture will not separate back into a liquid.
Procedure:Detail steps so anyone can reproduce this at school or home
Use a numbered list
|
Watch us in action!! |
THE SCIENCE BEHIND IT
Solutions: are see-through and the mixture does not separate after standing for any period of time. The particles are so small they cannot be separated by normal filtration.
Colloids: are mixtures with particle sizes that consist of clumps of molecules. The colloid looks consistent like fog or milk. The particles are large enough to scatter light. Colloids generally do not separate
on standing, and they are not separated by filtration.
Suspensions: are mixtures with even bigger particles than colloids.
The sizes of the particles are big enough so you can see them. Suspensions look muddy. They do not transmit light. Suspensions separate on standing. The mixture of particles can be separated by filtration.
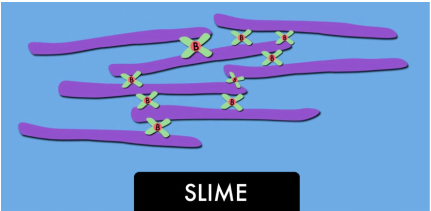
Slime is a colloid but, there is more to it than that! First it’s important
to understand how the molecules hold onto things. Most liquids,
like water, are made up of small, unconnected molecules that roll around each other. Single molecules are called monomers. In other materials, the monomers are connected together in matching,
repeating and making chains of molecules that are called polymers. Like spaghetti, they sort of roll over and around one another, but they are not connected to each other. The glue used in this activity
is a liquid polymer.
It’s like a box of steel chains that can slide across one another, each chain is made up of single links but the chains are not attached. Now, say you put magnets in with this box of chains. The magnets connect the chains together making a single blob of chains. If you reach in and try to grab one chain, you will lift out the entire pile.
Adding liquid starch to the glue and water does pretty much the same thing except through a chemical reaction. The starch, water and glue connect all the molecules together. When you pull it out you now have
a big blob of slime. The chemical reaction has made a slow-moving
form that is called a hydrogen-bonded, cross-linked polymer gel.
Conclusion
In conclusion, we reject our hypothesis. We thought that the slime would be a solution, but it was actually a colloid. A colloid is a mixture of particles that are clump sized. The colloid constantly looks foggy or milky. The particles of a colloid are large enough to scatter light. Unlike suspensions, colloids generally do not separate while standing or through filtration. Even though our hypothesis was wrong, the results could not be better. Lucky for you, because the slime is a colloid, it will never come apart. YAY! That means that you can have your slime forever as long as you do not leave it out because, then, it will dry out. Even a colloid can dry out because the water portion of it will evaporate. If your slime dries out you will lose the awesome slimy feeling. Remember, you can always make your own slime with the household ingredients of liquid starch, water, and glue.
Michael AmanteHi, my name is Michael. My favorite sport is baseball. I have a younger brother and older sister, three cats and a chinchilla!
|
Alex Kuchinsky
Hi, my name is Alex. My favorite sport is baseball, and I like basketball and football. Also, my favorite subject is math.
|
Andrew Palazzolo
Hi, my name is A.J. My favorite sport is basketball. My favorite subject is math. I am also the oldest child in my family.
|