Balloon Experiments:
Will Hot or Cold Be Bigger?
Question:
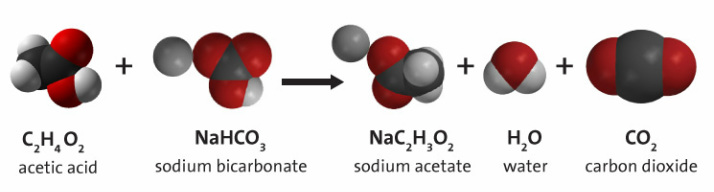
We know that combining an acid (vinegar) and base (baking soda) will create carbon dioxide, but will warming up the vinegar or cooling it off effect how big the balloon blows up?
Hypothesis:
Our prediction is yes. We think a change in the acid (vinegar) temperature will effect the reaction of the base (baking soda) and acid (vinegar). We think that a warmer acid will produce a greater reaction.
|
Materials
|
Safety ProceduresGlass Bottles: fragile/easily broken
Vinegar: keep away from eyes |
Procedure:
Special Needs: Close to a sink. |
Will hot, frozen, or room temperature vinegar make the biggest balloon?
|
The Science behind it
When you combine the baking soda and the vinegar it creates an acid-base reaction. The two chemicals work together to create a gas called carbon dioxide. The balloon expands because gasses need a lot of room to spread out and the carbon dioxide starts to fill the bottle, and then when it runs out of room it moves into the balloon to inflate it.
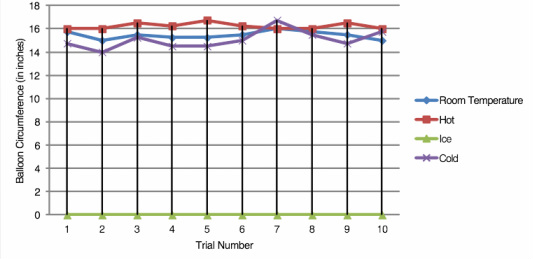
We put in the same amount of acid and base for each trial so each one should have produced the same amount of carbon dioxide and the same circumference of the balloon. The only variable we changed was the temperature of the vinegar. So a hotter acid produced a greater reaction.
We put in the same amount of acid and base for each trial so each one should have produced the same amount of carbon dioxide and the same circumference of the balloon. The only variable we changed was the temperature of the vinegar. So a hotter acid produced a greater reaction.
|
Room Temp. Vinegar
|
Hot Vinegar
|
Vinegar Ice cubes
|
Cold Vinegar
|
Conclusions
In conclusion, our experiment shows that changing the temperature of the vinegar (acid) will affect the circumference of the balloon.
When you heat the acid, the chemical reaction is greater resulting in a larger balloon circumference. When you chill the acid, the chemical reaction is lesser, resulting in a smaller balloon circumference.
We tested the vinegar with PH strips to see if the acidity changed in the vinegar when it was at different temperatures. It was the same.
When you heat the acid, the chemical reaction is greater resulting in a larger balloon circumference. When you chill the acid, the chemical reaction is lesser, resulting in a smaller balloon circumference.
We tested the vinegar with PH strips to see if the acidity changed in the vinegar when it was at different temperatures. It was the same.
About GregMy name is Greg. I am 10 years old. I play baseball and like to go skiing. My favorite subject is science.
|
About JoeyMy name is Joey. I am 10 years old. I love basketball and lacrosse. My favorite subject in school is math.
|
About Jack
My name is Jack. I am 11 years old. I play baseball, soccer and I am on a swim team. I like video games; my favorite game is "Clash of Clans". My favorite subject is math.
|