Can Crushers
Question:
Will a can implode if you heat it up and put it in cold water
Hypothesis:
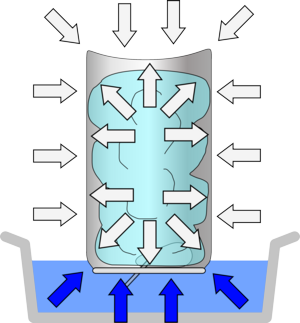
If a can is filled with a small amount of water, heated to the boiling point and then quickly inverted into a bowl of water, the sudden drop in vapor pressure will result in enough net inward pressure to quickly crush the can.
The Science behind it
A can is placed on a hot plate until steam rises, the water can be brought to the boiling point where its vapor pressure is equal to atmospheric pressure. The vapor will then push most of the air out of the can. If the can is quickly inverted into a beaker of water, sealing off the opening, the sudden drop in vapor pressure will result in enough net inward pressure to quickly crush the can.
Conclusions
Charlie Hopper
My name is Charlie, I have a brother and sister. I like to play sports - Football and Basketball and I like to dance to Gangman Style
|
Dylan Lanza
My name is Dylan and I play airsoft. I practice Judo and I have 2 dogs and 3 cats. I like to play Minecraft.
|